| [Ref.: #12433] |
Sample type/isolated from |
garden soil |
|
* marker position based on {}
|
|
Isolation sources categories |
| #Environmental |
#Terrestrial |
#Soil |
| #Engineered |
#Agriculture |
#Garden |
|
 |
-
Availability in culture collections
 External links External links
| [Ref.: #12433] |
Culture collection no. |
DSM 46092, ATCC 27624, IFO 13270, IMET 7649, JCM 3142, KCC A-0142, NBRC 13270, NRRL 2822, BCRC 12524, CGMCC 4.1144, CGMCC 4.1217, IMSNU 20060, KCTC 9428, VKM Ac-1244 |
| [Ref.: #82430] |
 SI-ID 92684 SI-ID 92684
|
* |
 |
Literature: |
|
Topic |
Title |
Authors |
Journal |
DOI |
Year |
|
| Phylogeny |
Amycolatopsis panacis sp. nov., isolated from Panax notoginseng rhizospheric soil. |
Peng G, Xiong DS, Li LC, Hu JY, Bao S, Chen YW, Li YQ, Xu LH, Miao CP, Zhao LX |
Int J Syst Evol Microbiol |
10.1099/ijsem.0.003202 |
2019 |
* |
| Phylogeny |
Amycolatopsis cihanbeyliensis sp. nov., a halotolerant actinomycete isolated from a salt mine. |
Tatar D, Sazak A, Guven K, Cetin D, Sahin N |
Int J Syst Evol Microbiol |
10.1099/ijs.0.050963-0 |
2013 |
* |
| Phylogeny |
Amycolatopsis jiangsuensis sp. nov., a novel endophytic actinomycete isolated from a coastal plant in Jiangsu, China. |
Xing K, Liu W, Zhang YJ, Bian GK, Zhang WD, Tamura T, Lee JS, Qin S, Jiang JH |
Antonie Van Leeuwenhoek |
10.1007/s10482-012-9823-y |
2012 |
* |
|
 References References-
| #12433 |
Leibniz Institut DSMZ-Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH ; Curators of the DSMZ;
DSM 46092
|
-
-
-
-
-
-
| #66794 |
Antje Chang, Lisa Jeske, Sandra Ulbrich, Julia Hofmann, Julia Koblitz, Ida Schomburg, Meina Neumann-Schaal, Dieter Jahn, Dietmar Schomburg:
BRENDA, the ELIXIR core data resource in 2021: new developments and updates.
Nucleic Acids Res. 49:
D498 - D508
2020 (
DOI 10.1093/nar/gkaa1025 , PubMed 33211880 )
|
-
| #67770 |
Japan Collection of Microorganism (JCM) ; Curators of the JCM;
|
-
| #68368 |
Automatically annotated from API 20E .
|
-
| #68379 |
Automatically annotated from API Coryne .
|
-
| #68382 |
Automatically annotated from API zym .
|
-
-
-
| #82430 |
Reimer, L.C., Lissin, A.,Schober, I., Witte,J.F., Podstawka, A., Lüken, H., Bunk, B.,Overmann, J.:
StrainInfo: A central database for resolving microbial strain identifiers
.
(
DOI 10.60712/SI-ID92684.1 )
|
- * These data were automatically processed and therefore are not curated
Change proposal
Successfully sent
|
|
Name and taxonomic classification

Morphology

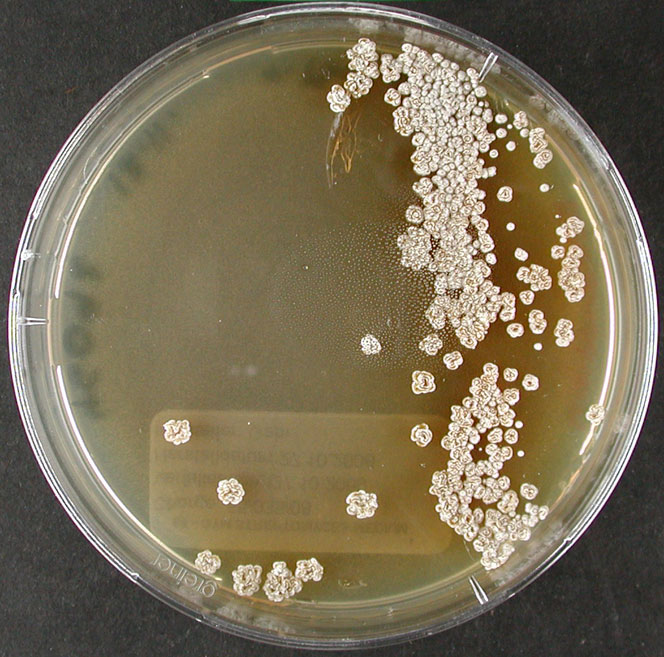
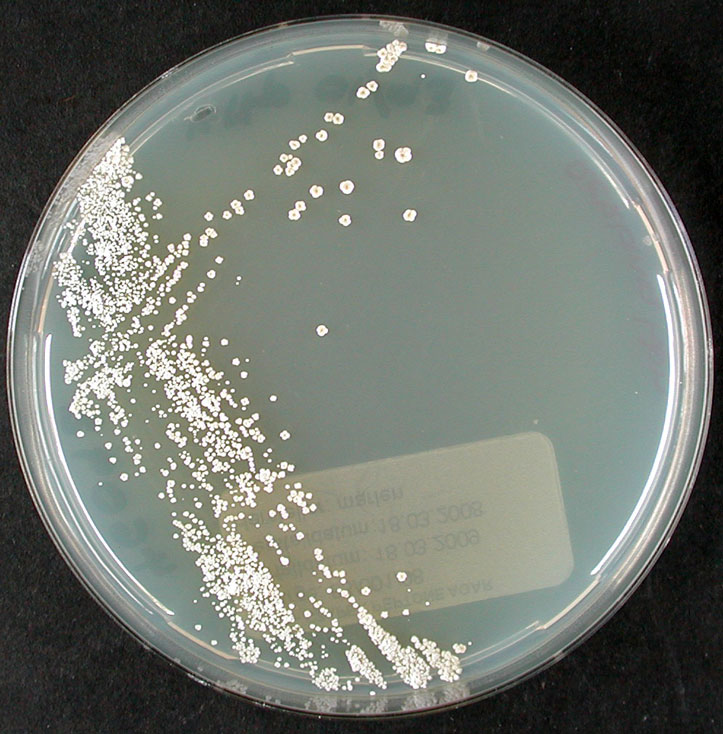
Culture and growth conditions

Physiology and metabolism

Isolation, sampling and environmental information

Safety information

Sequence information

Genome-based predictions

External links

References








