Streptomyces rochei DSM 41454 is a spore-forming, mesophilic bacterium that builds an aerial mycelium and was isolated from Soil.
- spore-forming
- mesophilic
- 16S sequence
- Bacteria
-
Information on the name and the taxonomic classification.
Name and taxonomic classification

|
-
Information on morphological and physiological properties
Morphology

-
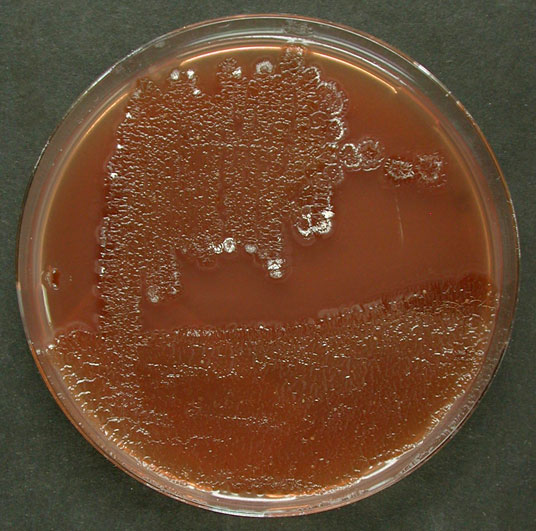
[Ref.: #10318] Caption: Medium 65 28°C [Ref.: #10318] Intellectual property rights: © Leibniz-Institut DSMZ
|
-
Information on culture and growth conditions
Culture and growth conditions

| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-
Information on physiology and metabolism
Physiology and metabolism

-
Information on isolation source, the sampling and environmental conditions
Isolation, sampling and environmental information

-
Information on possible application of the strain and its possible interaction with e.g. potential hosts
Safety information

-
Information on genomic background e.g. entries in nucleic sequence databass
Sequence information

-
Availability in culture collections
External links

References
-
#10318 Leibniz Institut DSMZ-Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH ; Curators of the DSMZ; DSM 41454 -
#18689 Wink, J.: Compendium of Actinobacteria. HZI-Helmholtz-Centre for Infection Research, Braunschweig . -
#20215 Parte, A.C., Sardà Carbasse, J., Meier-Kolthoff, J.P., Reimer, L.C. and Göker, M.: List of Prokaryotic names with Standing in Nomenclature (LPSN) moves to the DSMZ. IJSEM ( DOI 10.1099/ijsem.0.004332 ) -
#20218 Verslyppe, B., De Smet, W., De Baets, B., De Vos, P., Dawyndt P.: StrainInfo introduces electronic passports for microorganisms.. Syst Appl Microbiol. 37: 42 - 50 2014 ( DOI 10.1016/j.syapm.2013.11.002 , PubMed 24321274 ) -
#67770 Japan Collection of Microorganism (JCM) ; Curators of the JCM; -
#68368 Automatically annotated from API 20E . -
#69479 João F Matias Rodrigues, Janko Tackmann,Gregor Rot, Thomas SB Schmidt, Lukas Malfertheiner, Mihai Danaila,Marija Dmitrijeva, Daniela Gaio, Nicolas Näpflin and Christian von Mering. University of Zurich.: MicrobeAtlas 1.0 beta . - * These data were automatically processed and therefore are not curated
Change proposalYou found an error in BacDive? Please tell us about it! Note that changes will be reviewed and judged. If your changes are legitimate, changes will occur within the next BacDive update. Only proposed changes supported by the according reference will be reviewed. The BacDive team reserves the right to reject proposed changes. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||